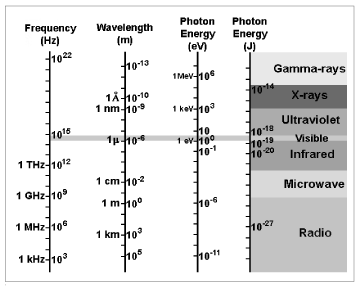
Wavelength (λ): The distance between two peaks of a cycle. A Watt is a Joule (1 J) of work done in a second (1 s). Volt (V): A potential due to an electric field. Time (t): The symbol for time in seconds (s). Ohm (Ω): A measure of resistance or impedance. The distance light travels in 1/299,792,458 second. Length: (l): The symbol for distance in meters (m). 
Joule (J): The work required to move a Newton (1 N) a meter (1 m). Horse Power (HP): The power a horse can achieve. One Farad equals a capacitor that has a Coulomb (1 C) of charge on it with a voltage separation of a Volt (1 V). The unit cps (or kilocycles, megacycles, etc.) is more often seen in older documents.įarad (F): The SI unit for Capacitance (C). This is equivalent to Hertz, but Hertz is the official unit. Equals a coulomb (1 C) of charged particles moving past a point in one second (1 s).Ĭycles per second (cps): As the name implies, a measurement of frequency in full cycles of a wave per second. Hertz, meters, and seconds are units.Īmpere, Amp (A): The SI unit for current I. So we should copy all of these to the definitions page as well? Things like frequency, length, time and wavelength are not units they should be in the definitions section and not here. Ideally all the definitions in Units should find their way into the main definitions page. That's where the units page comes in handy as you only have to search through a small number of units to find the unit you want. Imagine doing a math problem and coming across Ξ. My take on the units page is that it is a guide for doing math problems. In equations, wavelength is indicated using the Greek letter lambda (). The distance between one crest (or trough) of one wave and the next is the wavelength of the wave. (Do we want these separated from the definitions page? or should we include all of these on the definitions page and just have a separate units page too?) The wavelength is a property of a wave that is the distance between identical points between two successive waves. One reason people don't understand math or Physics very well is that it literally is Greek to them. The majority of these units are named after famous people in the field who either discovered the units or had someone else name the units after them. When there are shorter wavelengths they are expressed as negative exponential. In the case of light, the frequency, symbolized by the Greek letter nu (), of any wave equals the speed of light, c, divided by the wavelength : thus c. Notably, exponential powers of 10 are used when wavelengths are of a large property. wavenumber, also called wave number, a unit of frequency, often used in atomic, molecular, and nuclear spectroscopy, equal to the true frequency divided by the speed of the wave and thus equal to the number of waves in a unit distance. While measuring wavelength the multiples or fractions of a metre is also used. Since absorption, \(\epsilon\), and path length are known, we can calculate the concentration \(c\) of the sample.These are the units you would find when doing problems in electronics. The SI unit of wavelength is metre usually denoted as m. Because a standard spectrometer uses a cuvette that is 1 cm in width, \(l\) is always assumed to equal 1 cm. Search for crossword clues found in the Daily Celebrity, NY Times, Daily Mirror, Telegraph and major. The path length is measured in centimeters. Answers for wavelength unit crossword clue, 8 letters. As a result, \(\epsilon\) has the units: L Since absorbance does not carry any units, the units for \(\epsilon\) must cancel out the units of length and concentration. The molar extinction coefficient is given as a constant and varies for each molecule. \(\epsilon\) is the molar extinction coefficient or molar absorptivity (or absorption coefficient),.\(A\) is the measure of absorbance (no units),.For the relationship to hold mathematically, if the speed of light is used in m/s, the wavelength must be in meters and the frequency in Hertz. For this reason, Beer's Law can only be applied when there is a linear relationship. As the wavelength of a wave increases, its frequency decreases. Figure 5: Transmittance (CC BY-4.0 Heesung Shim via LibreTexts)īeer-Lambert Law (also known as Beer's Law) states that there is a linear relationship between the absorbance and the concentration of a sample. You need to know about these quantities used in waves - what they mean, the symbols used for them and the units used to measure them. The length \(l\) is used for Beer-Lambert Law described below. Figure 5 illustrates transmittance of light through a sample. With the amount of absorbance known from the above equation, you can determine the unknown concentration of the sample by using Beer-Lambert Law. Where absorbance stands for the amount of photons that is absorbed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
